Abstract
Purpose: Despite the success of CAR T-cell therapy in pts with relapsed/refractory (R/R) DLBCL, some pts still fail treatment, and their prognosis is dismal. Our aim is to elucidate factors that govern the clinical performance of CAR T therapy given as standard of care (SOC) for R/R DLBCL at a single institution and to recognize the group of pts who are at risk for relapse after CAR T as early intervention may help improve the outcomes.
Methods: We performed a retrospective study of R/R DLBCL pts treated with CAR T-cell as SOC at our center between Jan 2018 and Feb 2021. Disease characteristics, ferritin and CRP levels were collected pre-treatment initiation; CBC data were collected at time of apheresis. The study was IRB-approved at our center.
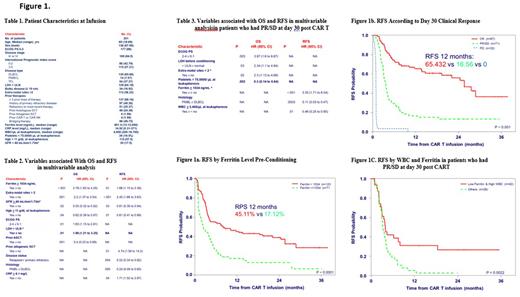
Results: Characteristics of the pts at pre-conditioning for CAR T are listed in Table 1. Of 201pts, 190 (94.5%) received axicabtagene ciloleucel, and 11 (5.5%) received tisagenlecleucel. Median time from apheresis to infusion was 29 days (range, 20-111). Regarding adverse events of special interest, 6% had cytokine release syndrome of > grade 3 and 38.8% had neurotoxicity > grade 3. Day 30 death rate was 4.5%. Nine (4.68%) pts developed AML/MDS and 3 (1.56%) additional pts had prolonged pancytopenia. Sixty (29.8%) pts died post Day 30 [42 of progressive disease (PD); PD/AML =3; PD/Infection =2; PD/Toxicity =2; Infection =9; unknown=2). Four pts lost follow-up after PD. With a median follow-up from infusion of 16.86 (0.92-38.7) months, the 12-months overall survival (OS) rate was 55.48 %(95%CI:49-63); 12-months relapse-free survival (RFS) was 34.57% (95% CI: 28-42).
In multivariate analysis, the factors with independent influence on both RFS and OS were serum ferritin, > 2 extra-nodal sites of disease involvement, glomerular filtration rate pre-conditioning and Hb level at apheresis (Table 2, Figure 1a). Salvage therapies were received by 38 pts with PD after CAR T (median 1, range 1-6 lines of therapies), including 9 pts who received an allogeneic stem cell transplant. Only 5 (13%) of these 38 pts achieved CR. Therefore, we assessed response by day 30 to predict which pts were at risk for relapse prior experiencing an actual progression. We found a significant difference in OS and RFS in patients who were in CR vs PR/SD after CAR T. The 12-months OS rates were 75.52% and 54.38%, respectively (P<0.001). The RFS rates were 65.4% vs 16.56%, respectively (P<0.001, Figure 1b). In multivariate analysis, the factors with independent influence on RFS in Day 30 PR/SD pts were serum ferritin level pre-conditioning, histology (primary mediastinal vs DLBCL), and WBC at apheresis (figure 1c).
Conclusions: Predictors of outcomes after CAR T cell therapy are multifactorial. However, we found that simple clinical tools by Day 30 (pts not achieving CR by imaging, ferritin level pre-CAR T infusion, WBC at apheresis) are associated with outcomes. Studies are needed to study whether integration of CAR T with innovative therapies in such high-risk patients may improve survival.
Disclosures
Neelapu:Bristol Myers Squibb: Consultancy, Honoraria, Other: Personal fees, Research Funding; Novartis: Consultancy, Honoraria, Other: Personal fees; Bio Ascend: Consultancy, Honoraria; Legend Biotech: Consultancy, Honoraria, Other: Personal fees; Precision Biosciences: Consultancy, Honoraria, Other: Personal fees, Research Funding; Aptitude Health: Consultancy, Research Funding; Cell Medica/Kuur: Consultancy, Honoraria, Other: Personal fees; Merck: Consultancy, Honoraria, Other: Personal fees, Research Funding; Kite: Consultancy, Honoraria, Other: Personal fees, Research Funding; Medscape: Consultancy, Honoraria; Incyte: Consultancy, Honoraria, Other: Personal fees; Bluebird Bio: Consultancy, Honoraria; Unum Therapeutics: Consultancy, Honoraria, Other: Personal fees, Research Funding; Adicet Bio: Consultancy, Honoraria, Other: Personal fees, Research Funding; Calibr: Consultancy, Honoraria, Other: Personal fees; Allogene Therapeutics: Consultancy, Honoraria, Other: Personal fees, Research Funding; Celgene: Consultancy, Honoraria, Other: Personal fees, Research Funding; Pfizer: Consultancy, Honoraria, Other: Personal fees; Takeda Pharmaceuticals: Patents & Royalties: related to cell therapy.; Poseida: Research Funding; Cellectis: Research Funding; Karus Therapeutics: Research Funding; Acerta: Research Funding. Ahmed:Chimagen: Consultancy, Research Funding; Xencor: Research Funding; Myeloid Therapeutics: Consultancy; Servier: Membership on an entity's Board of Directors or advisory committees; Seagen: Research Funding; Merck: Research Funding; Tessa Therapeutics: Consultancy, Research Funding. Kebriaei:Ziopharm: Research Funding; Pfizer: Consultancy; Amgen: Research Funding; Kite: Consultancy; Jazz: Consultancy. Westin:MorphoSys/Incyte Corporation: Consultancy, Research Funding; Bristol Myers Squibb: Consultancy, Research Funding; AstraZeneca: Consultancy, Research Funding; Merck: Consultancy; Iksuda: Consultancy; Kite, a Gilead Company: Consultancy, Research Funding; Abbvie/GenMab: Consultancy; MonteRosa: Consultancy; Calithera: Consultancy, Research Funding; ADC Therapeutics: Consultancy, Research Funding; Genentech/Roche: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; SeaGen: Consultancy. Shpall:Fibroblasts and FibroBiologics: Consultancy; NY blood center: Consultancy; Navan: Consultancy; axio: Consultancy; adaptimmune: Consultancy; Bayer: Honoraria; Takeda: Patents & Royalties; Affimed: Other: License agreement. Champlin:Actinium: Consultancy; Johnson &Johnson: Consultancy; Omeros: Consultancy; Kadmon: Consultancy; General Oncology: Other: Data Safety Monitoring Board; Bluebird: Other: Data Safety Monitoring Board; Cell Source Inc.: Research Funding. Nastoupil:Genentech/Roche, MEI, Takeda: Other: DSMC; ADC Therapeutics, BMS, Caribou Biosciences, Epizyme, Genentech/Roche, Gilead/Kite, Genmab, Janssen, MEI, Morphosys, Novartis, Takeda: Honoraria; BMS, Caribou Biosciences, Epizyme, Genentech, Gilead/Kite, Genmab, Janssen, IGM Biosciences, Novartis, Takeda: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal